Abstract
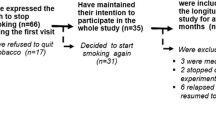
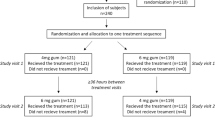
There were two experiments on abstinence from smokeless tobacco. The purpose of the first experiment was to determine abstinence effects from smokeless tobacco. The purpose of the second experiment was to examine the effects of different doses of nicotine gum on smokeless tobacco abstinence effects. The subjects were male Copenhagen smokeless tobacco users who underwent 3 days of baseline measurement while continuing to use smokeless tobacco ad libitum, and 5 days of the experimental condition. In the first experiment, the subjects were assigned randomly to one of two groups and compared: continuous smokeless tobacco users (n=10), and deprivation plus no nicotine gum (n=10). In the second experiment, subjects were assigned randomly and in a double-blind fashion to one of three groups and compared: (1) deprivation plus 0 mg nicotine gum (n=20); (2) deprivation plus 2 mg nicotine gum (n=20); and (3) deprivation plus 4 mg nicotine gum (n=20). The first experiment showed significant increases upon abstinence for the following variables: (1) craving; (2) difficulty concentrating; (3) restlessness; (4) excessive hunger; (5) eating; (6) reaction time; (7) variability of reaction time and (8) total withdrawal scores for both the self-rated and the observer-rated forms. The second experiment showed that nicotine gum failed to significantly reduce smokeless tobacco abstinence effects, although those with high cotinine levels may receive some benefit from nicotine gum.
Similar content being viewed by others
References
Abrams DB, Follick MJ, Biener L, Carey KB, Hitti J (1987) Saliva cotinine as a measure of smoking status in field settings. Am J Public Health 77:846–848
Benowitz NL, Kuyt F, Jacob P, Jones RT, Osman AL (1983) Cotinine disposition and effects. Clin Pharmacol Ther 34:604–611
Benowitz NL, Porchet H, Sheiner L, Jacob P III (1988) Nicotine absorption and cardiovascular effects of smokeless tobacco use: comparison with cigarette and nicotine gum. Clin Pharmacol Ther 44:23–28
Benowitz NL, Jacob P III, Yu L (1989) Daily use of smokeless tobacco: systematic effects. Anna Int Med 111:112–116
Carskadon MA, Dement WC, Mitler MM, Guilleminault MD, Zarcone VD, Spiegel R (1976) Self-report vs. sleep lab findings in 122 drug-free subjects with complaints of chronic insomnia. Am J Psychiatry 133:82–88
Christen AG, McDaniel RK, Doran JE (1979a) Snuff dipping and tobacco chewing in a group of Texas college athletes. Tex Dent J 97:6–10
Christen AG, Armstrong WR, McDaniel RV (1979b) Intraoral leukoplakia, periodontal breakdown, and teeth loss in a snuff dipper. J Am Dent Assoc 98:584–586
Cummings SR, Richard RJ (1988) Optimum cutoff points for biochemical validation of smoking status. Am J Public Health 78:574–575
Goldsmith DF, Winn DM (1980) Hazards of snuff. Lancet 1:825
Greer RO, Poulson TC (1983) Oral tissue alterations associated with the use of smokeless tobacco by teenagers. Oral Surg 56:275–284
Gritz ER, Baer-Weiss V, Benowitz NL, Van Vanakis H, Jarvik ME (1981) Plasma nicotine and cotinine concentrations in habitual smokeless tobacco users. Clin Pharmacol Ther 30:201–209
Gross J, Stitzer ML (1989) Nicotine replacement: ten week effects on tobacco withdrawal symptoms. Psychopharmacology 98:334–341
Hall SM, Tunstall C, Ginsberg D, Benowitz NC, Jones RT (1987) Nicotine gum and behavioral treatment: a placebo controlled trial. J Consult Clin Psychol 55:603–605
Hanter SM, Webber LS, Berenson GS (1980) Cigarette smoking and tobacco usage behavior in children and adolescents: Bogalusa heart study. Prev Med 9:701–712
Harper S (1980) In tobacco, where there's smokeless fire. Advertising Age 51:85
Hatsukami DK, Hughes JR, Pickens RW, Svikis D (1984) Tobacco withdrawal symptoms: an experimental analysis. Psychopharmacology 84:231–296
Hatsukami DK, Gust SW, Keenan RM (1987) Physiologic and subjective changes from smokeless tobacco withdrawl. Clin Pharmacol Ther 41:103–107
Hatsukami DK, Dahlgren L, Zimmerman R, Hughes JR (1988) Symptoms of tobacco withdrawal from total cigarette cessation versus partial cigarette reduction. Psychopharmacology 94:242–247
Hjalmarson AIM (1984) Effects of nicotine chewing gum in smoking cessation. A randomized, placebo-controlled, double-blind study. JAMA 252:2835–2838
Hughes JR, Hatsukami DK (1986) Signs and symptoms of tobacco withdrawal. Arch Gen Psychiatry 43:289–294
Hughes JR, Hatsukami DK, Pickens RW, Krahn D, Malin S, Luknic A (1984) Effect of nicotine on the tobacco withdrawal syndrome. Psychopharmacology 83:82–87
Jarvis MJ, Raw M, Russell MAH, Feyerabend C (1982) Randomized controlled trial of nicotine chewing-gum. Br Med J 285:537–540
Keenan RM, Hatsukami DK, Anton DJ (1989) The effects of smokeless tobacco deprivation on performance. Psychopharmacology 98:128–130
Kornitzer M, Kittel F, Dramaix M, Bourdoux P (1987) A double blind study of 2 mg versus 4 mg nicotine-gum in an industrial setting. J Psychosom Res 31:171–176
Lichtenstein E, Severson HH, Friedman LS, Ary DV (1984) Chewing tobacco use by adolescents: prevalence and relation to cigarette smoking. Addict Behav 9:351–355
Massey JD Jr, Moore GF, Yonkers AJ (1984) Smokeless tobacco: a risk factor in oral cancer. Ear Nose Throat J 63:453–458
Mattson ME, Winn D (1989) Smokeless tobacco: association with increased risk for cancer. In National Cancer Institute Res Monograph No 8, US Government Printing Office, Washington DC, NIH publication no. 89–3055, pp 13–16
McNair DM, Lorr M, Droppleman LF (1971) Manual for the profile of mood states. Educational and Industrial Testing Service, San Diego
Morse RM, Norvich RC, Graf JA (1977) Tobacco chewing. An unusual case of drug dependence. Mayo Clin Proc 52:358–360
Office of Medical Applications of Research (1986) NIDA: Health applications of smokeless tobacco use. JAMA 255:1045–1048
Poulson TC, Lindenmuth JE, Creer RO (1984) A comparison of the use of smokeless tobacco in rural and urban teenagers. CA 34:248–261
Schaefer SD, Henerson AH, Glover ED, Christen AG (1985) Patterns of use and incidence of smokeless tobacco consumption in school age children. Arch Otolaryngol 111:639–642
Sepkovic DW, Haley NJ (1985) Biomedical applications of cotinine quantitation in smoking related research. Am J Public Health 75:663–665
Stookey GK, Katz BP, Olson BL, Drook CA, Cohen SJ (1987) Evaluation of biochemical validation measures in determination of smoking status. J Dent Res 66[10]:1597–1601
Tonnesen P, Fryd V, Hansen M, Helsted J, Gunnersen AB, Forchammer H, Stocker M (1988) Effect of nicotine chewing gum in combination with group counseling on the cessation of smoking. N Engl J Med 318:15–18
US Department of Health and Human Services (1986) The health consequences of using smokeless tobacco. US Government Printing Office. DHHS publication no. 86–2874
US Department of Health and Human Services (1988) The health consequences of smoking: nicotine addiction. A report of the surgeon general. US Government Printing Office. DHHS publication no. 88–8406
Yellin AMA (1980) Standard visual stimulus for use in studies of attention and attention deficit disorders: toward the development standardized and selective attentions tests. Res Commun Psychol Psychiatr Behav 5:137–143
Author information
Authors and Affiliations
Additional information
Sponsored by the National Institute on Drug Abuse Research Grant No. DA05013
Rights and permissions
About this article
Cite this article
Hatsukami, D., Anton, D., Keenan, R. et al. Smokeless tobacco abstinence effects and nicotine gum dose. Psychopharmacology 106, 60–66 (1992). https://doi.org/10.1007/BF02253589
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02253589